CARBON DIOXIDE GAS
1. Write down the molecular formula and molecular weight of carbon dioxide.
The molecular formula of carbon dioxide is CO2 and its molecular weight is 44 a.m.u.
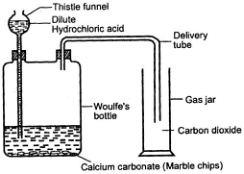
2. How is carbon dioxide prepared in laboratory?
In laboratory carbon dioxide gas is prepared by the reaction of dilute hydrochloric acid and calcium carbonate.
Chemical equation:
CaCO3 + 2HCl(dil) → CaCl2 + H2O + CO2
3. What will happen if the dilute hydrochloric acid is replaced by dilute sulphuric acid?
When dilute sulphuric acid is used, calcium sulphate is formed which covers the marble and prevents further reaction.
CaCO3 + H2SO4(dil) → CaSO4 + H2O + CO2
Tests for CO2:
- Extinguishes burning splint
- Turns lime water milky: Ca(OH)2 + CO2 → CaCO3↓ + H2O
Uses of CO2:
- Photosynthesis
- Fire extinguishers
- Carbonated drinks
- Dry ice refrigeration
AMMONIA GAS (NH3)
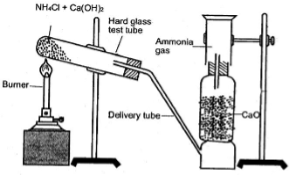
Laboratory Preparation:
2NH4Cl + Ca(OH)2 → CaCl2 + 2H2O + 2NH3
Industrial Preparation (Haber Process):
N2 + 3H2 ⇌ 2NH3 (450°C, 200 atm, Fe/Mo catalyst)
Tests for NH3:
- Turns moist red litmus blue
- Pungent smell
- Forms white fumes with HCl: NH3 + HCl → NH4Cl
Uses of NH3:
- Fertilizer production
- Plastics manufacturing
- Cleaning products
- Refrigeration
Important Chemical Compounds
| Common Name | Chemical Name | Formula |
|---|---|---|
| Lime Water | Calcium Hydroxide | Ca(OH)2 |
| Limestone | Calcium Carbonate | CaCO3 |
| Quick Lime | Calcium Oxide | CaO |
Ammonia Fountain Experiment
Demonstration:
- Beaker filled with water + red litmus
- Top flask filled with NH3 gas
- Press the dropper
Observation: Water rushes up, solution turns blue
Explanation: NH3 dissolves creating vacuum, solution becomes basic




0 Comments